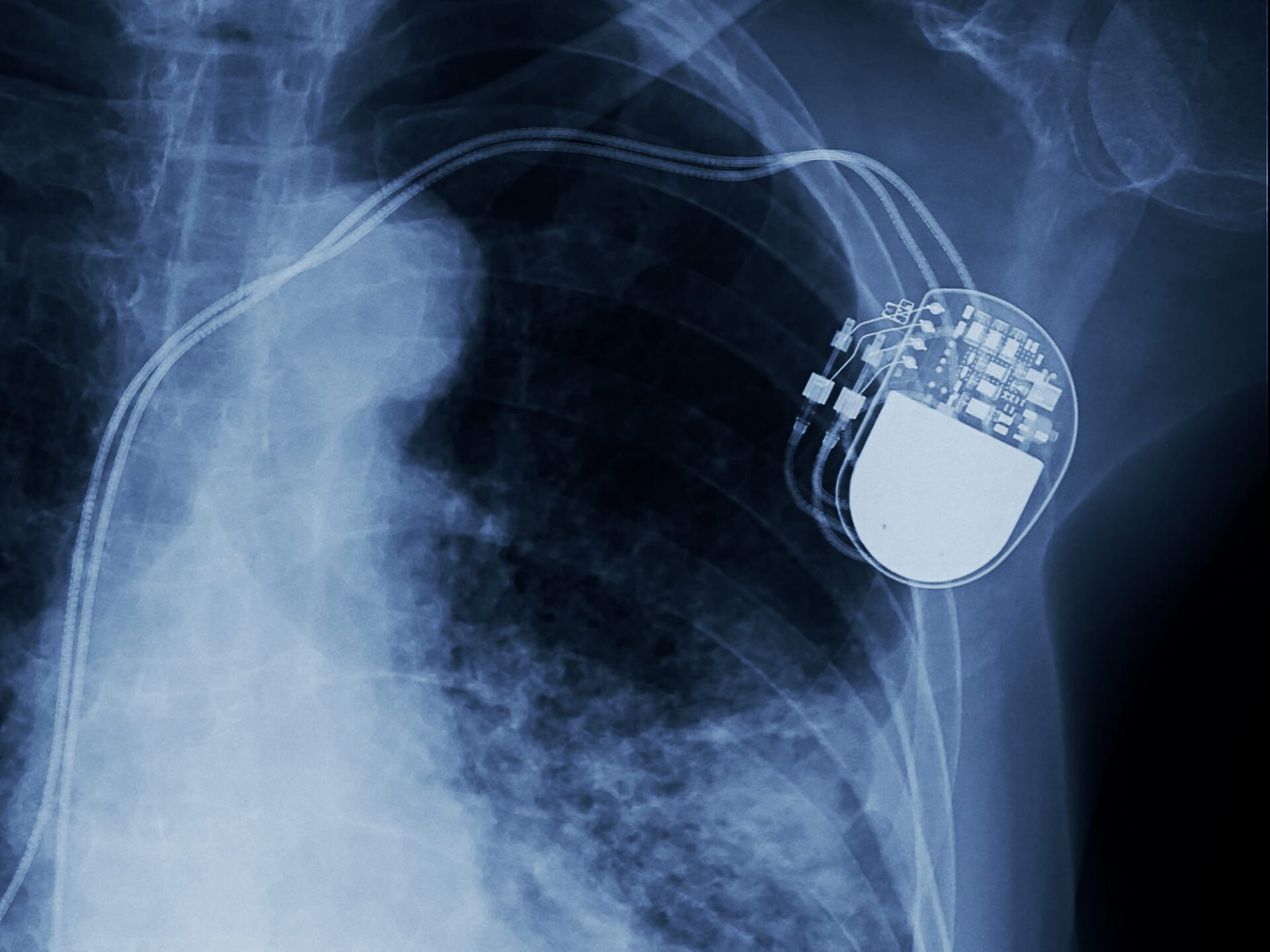
The nature of implanted medical devices means that ensuring their integrity is critically important. A Quality Magazine article, titled “Nondestructive Testing of Implanted Medical Device Materials,” examines the problems that patients could experience if an implanted medical device fails while in use. The article details how not taking proper precautions can result in serious consequences for the device manufacturer. Testing the materials used for manufacturing implanted medical devices and the actual medical devices themselves to ensure integrity is extremely important.
Ensuring Integrity With Nondestructive Testing
Nondestructive testing is an important part of ensuring implanted medical device integrity. The article, written by Rich Goodwin and Phil Trach, discusses two types of nondestructive testing commonly used to detect defects in materials and finished parts without causing damage to the samples. The step-by-step testing processes are explained in detail for two specific test methods, Liquid Penetrant Testing and Ultrasonic Testing, which reveal surface and/or subsurface defects with reliable accuracy.
Although the article was written about medical implants, the basic principles and test methods are relevant to any industry where “high-risk” or “mission-critical” products are manufactured. For example, certain parts manufactured for aircraft, automobiles, and military applications could also cause devastating results if they do not perform as expected. Nondestructive testing is a very effective solution for ensuring product integrity for manufacturers and end users in many industries.
Quality Magazine is a monthly publication that serves the quality assurance and process improvement needs of manufacturing professionals.
If you need an experienced, quality-oriented laboratory, contact LTI for a quote.
Related Articles
How Small a Defect Can UT Find
Customers often ask LTI how small a defect ultrasonic testing (UT) can find. This question is very difficult to answer…
Ethics in Laboratory Testing
What could be more important than ethics in the world of materials testing? Testing laboratories have an obligation to adhere…
Quality Considerations in Chemical Analysis
A Well-Planned and Managed Quality Control Program When you need chemical analysis services, you expect to receive accurate testing and…
Spectroscopy Explained
In our new article titled “Spectroscopy Explained”, you’ll learn how chemical testing is performed to answer many questions about material…
Third-Generation Family Member Becomes President
Passing the Baton After 25 years as president of Laboratory Testing Inc. (LTI), Michael J. McVaugh is retiring from the…
Promoting NDT to the Next Generation
Nondestructive Testing (NDT) helps ensure the reliability and safety of materials and products manufactured by many industries, yet it’s a…